Neuroscience
Summer 2018-Current
Summer of 2018, I joined Dr. Watson’s lab as an incoming first year to immerse into the research environment (ARC Program). Before coming to W&L, I never thought that I would have the opportunity to join a research lab, nevertheless, a neuroscience lab. My scholar’s journey was slow but steady, gradually gaining the trust of Dr. Watson by doing small tasks that seemed mundane. Because of this experience, I decided to major in neuroscience and continued to conduct research in Dr. Watson’s lab for the next four years. I am very thankful for this experience. My investigation into the optic nerve regeneration in the frog model will be discussed below.
The Frog Squad

Curious
Collaborative and Teamwork
Inspiring
meticulous
Resourceful
The Objective
The objective of our lab is to study the optic nerve (ON) regeneration process in the Xenopus Laevis model. The African Clawed Frog has the ability to regenerate their optic nerves following an injury. while mammals cannot We hope to gain insight on the regeneration process by looking at the molecular level, focusing on the protein UCHL1 which is important in the ubiquitin pathway. Two current theories of how the ON regrows are 1) injured RGCs must die and then be replaced by de novo RGCs or (2) injured RCGs are protected from cell death and then regrow their axons. We hope to apply our understanding in the animal model to gain insight on the regeneration process of the CNS in humans.
The Problem
One of the problem we have is how to standardize ON injuries. Previously, the old ONC surgery is done by a surgical incision made in the roof of the buccal cavity. However, this method is difficult to do and potentially causes bleeding. One of the main problem in our lab is developing a optic nerve crush (ONC) surgery procedure that would ensure 100% accuracy of crushing the ON, avoid injuring the blood supply nearby, and guarantee a quick recovery post-surgery.
The Solution
The solution that we come up with is developing a new ONC surgery procedure through the back of the eye. Instead of going through the roof of the buccal cavity, we perform an incision around the cornea of the eye which allow us to have greater visual field when crushing the optic nerve. The new method allows for 100% of verifying the crush doing ONC surgery. In addition, we are able to distinguish and avoid injuring the blood supply that runs side by side the optic nerve. Lastly, frogs are able to recover quickly post-surgery because no blood supply is damaged with a near 100% survival rate. I help to develop the technique by designing a mini experimental trials to find the best surgery procedure. I perform different techniques and collect results on the accuracy of the crush and survival rate.
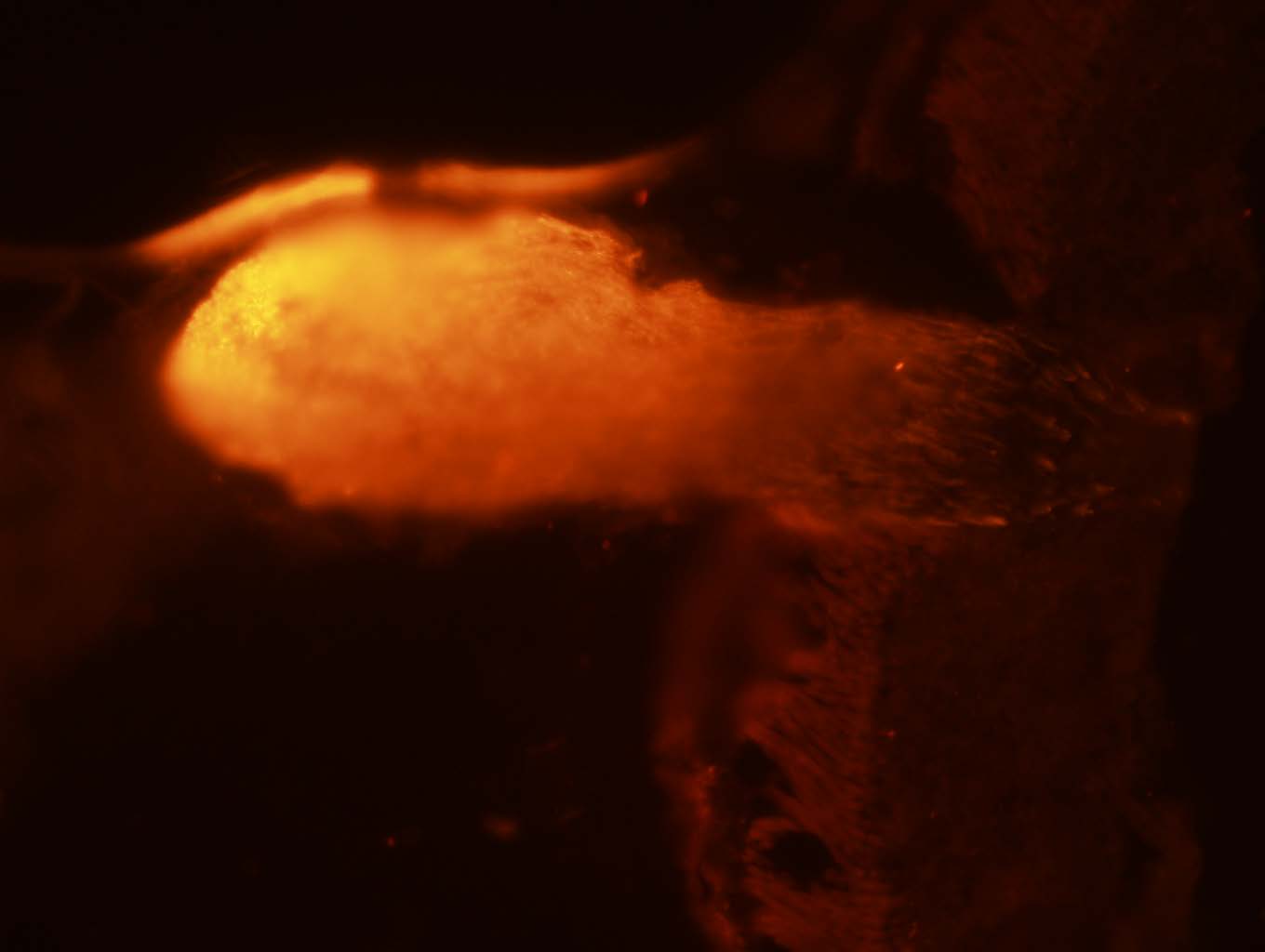
(1) An optic nerve head that has been stained with sncg antibody.
Note: this is not the best image I have taken. Because of copyright, I do not have access to current data or published images.
Imaging Results
After the surgery, we collect the tissues to analyze the level of protein expressions at the crush site. We carefully dissect out the eye and optic nerve to not disrupt the ONC site. Afterward, we slice the tissues into thin sections (around 7micrometers). We do antibody staining to visualize the target proteins and signaling molecules. Moreover, we image the tissues using the confocal microscope and the fluorescence microscope.
Surgery and Dissection
As I have mention earlier, I contribute to develop a new surgery procedure in lab. I conducted mini experiment to find the best method to do surgery. In addition, I also carefully dissect out the eyes, optic nerves, and brains without damaging the tissues. I have been doing it throughout my years of college.
- Surgery and Dissection 90%
Slicing and Staining
I can proficiently slice beatiful sections of the retina and optic nerves. A main part of working in lab is developing sectioning skills on the cryostat. In addition, I am able to do staining on collected tissues. I can follow procedure precisely to get a quality staining to image.
- Slicing and Staining 80%
Imaging and Data Analysis
I have to most experience with imaging the retina and optic using the fluoroscope. I am able to independently navigate the setting and levels to get the best acurrate image. I have branch out and learn to use the confocal microscope. On data analysis, I have taken classes to help me analyze data. I will be working with Dr. Toporikova to learn more about using Python coding language to analyze biological data.
- Imagaing and Data Analysis 85%
W&L History
Fall 2018
In the fall of my first year, I conducted research on the history of W&L under the guidance of Professor Wilson for my writing class. The research that my group did was on W&L history on coeducation and integration of minorities on campus. We narrow our research to focus on the integration of color women onto W&L campus around the early 1970s. Our methodologies include data mining in the University’s archive, looking through the yearbooks and the University’s paper Ring Tum Phi, and conducting interviews of current students who their mothers or sisters were among the first women of color on W&L campus. We created a visual essay that was contributed to the University’s archive.
To watch the video below, please click on the link below!
https://wlu.box.com/s/kcfoxfxjzov5i962xjx01a4obq8cntlq
Biology & Computer Science
Summer 2020
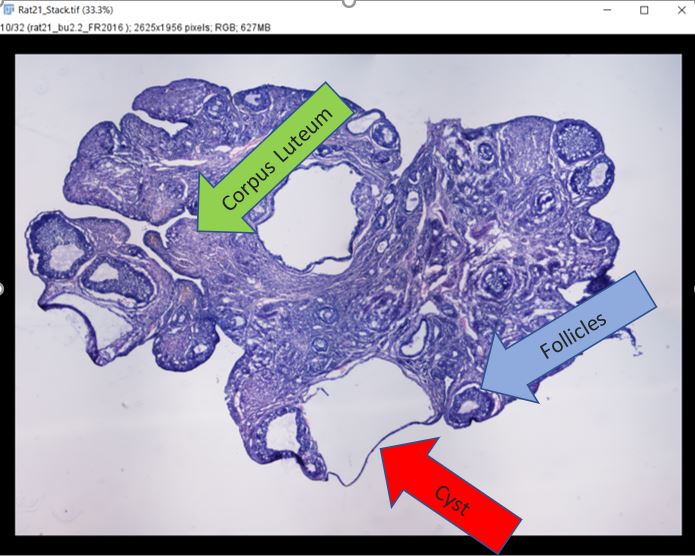
Because of COVID-19, my summer plan was canceled. However, I connected with a Dr. Toporikova from the Biology department at W&L. This research opportunity was perfect since it could be safely done at home. My project this summer was to build a computational algorithm to classify different structures of the rat’s ovaries. The goal is to design a computer learning code that will help researcher distinguish between follicles, cysts, and corpus luteum without any bias. My background was in neuroscience with little coding experience. However, this set back did not stop me from doing what I love, research. For 14 weeks, I learned how to code in Python to an intermediate proficiency, using my new skills to process images and data collections.
The project is divided into two parts: 1) collecting images of follicles, cysts, and corpus luteum and 2) write a computer learning algorithm to recognize the three different structures. For the first part, I must learn to distinguish the three structures myself which came with practice. The process was slow but I eventually gain a new skill to analyze tissues samples and pull out relevant features. Toward the end of summer, I was chosen by my PI to mentor a new student. I taught her to identify the different ovaries’s structures. This is a very rewarding experience for me because it teaches me to be a better communicator and mentor while showing me how much I have progress in 10 weeks. The second part of research was to build a code that would do all the work that people could do. Using my new skill in Python, I work with my team to resize images, put filters on them, sort them into csv files, and ultimately run a code on the collected images to classify the structures. We have gotten 99% accuracy when identifying cyst versus corpus luteum. In the future, we hope to imporve the accuracy for identifying cysts and follicles since there were lots of similarity between them. We also hope to write a code that will distinguish between all three structures.
Skills Obtain From Research
- Coding in Python 80%
- Analyzing Tissues 90%
- Working with ImageJ 85%
- Communication using online platform 95%
- Time Management 90%
- Team Work 90%
